First-in-world cancer trials at The Alfred
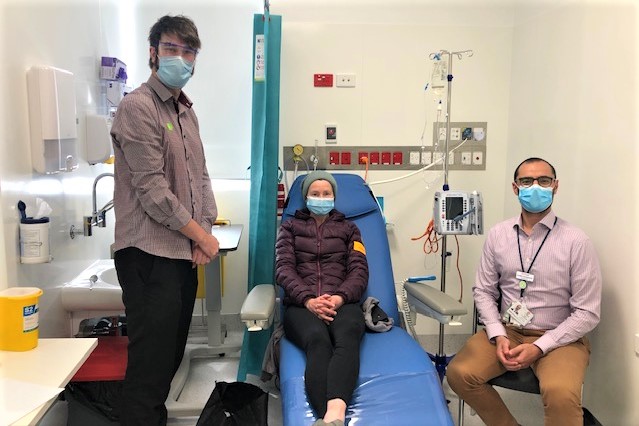
Alfred Cancer Trials (ACT) has begun testing two new Phase 1 cancer drugs, treating the first patients in the world on these trials.
These first-in-human drug trials for advanced cancers are currently at the safety testing stage, to determine the best dose for patients.
The Alfred has been selected as one of only a few sites permitted to run these Phase 1 cancer trials. For patients like Liza Pharaoh, these trials offer hope when traditional treatment options have been exhausted.
Liza has advanced colorectal cancer and travels regularly from Gippsland to receive treatment with the trial drug.
ACT Research Manager Cheryl-Ann Hawkins said these trials are a milestone for ACT.
"To have the opportunity and support of our sponsors to manage the first in the world dosing of not one, but two, anti-cancer drugs, is remarkable,” she said.
ACT has been conducting first-in-human drug trials since 2019 and has developed an expert team of medical oncologists and registered nurses to support the growth of these complex drug trials. The team will have 16 early phase trials open to recruitment by the end of October 2020.

_500_333.jpg)